
Key recommendations in the report include normalizing mask wearing and avoiding the supply chain failures.

Key recommendations in the report include normalizing mask wearing and avoiding the supply chain failures.

A new study supports the conclusion that the “brain fog” reported by many who have recovered from mild COVID-19 infection is real and has an anatomical basis.

Compliance has increased up to 50% on key infection prevention and control practices at a South Carolina health system due to these short emoji-cartoons.

Developers designed a game to teach infection prevention during COVID-19 to senior nursing students.

Dr. Kevin Kavanagh shares his perspective on the new CDC mask guidance.

Investigators used 3 behavior models to study how to increase health care workers’ compliance with infection prevention and control practices.

Take 5 minutes to catch up on Infection Control Today’s highlights for the week ending February 4.

A public/private enterprise taps into the entrepreneurial urge that will hopefully help the United States avoid the disastrous shortages of personal protective equipment that deviled the health care system in the first months of the COVID-19 pandemic.

ICU nurses, more than other clinicians, because they have the most interaction with patients. Non-clinical workers because they may not be knowledgeable enough about mitigation methods, a study states.
Reinfections among study participants most likely occurred outside the hospital setting where workers might let their guard down and not practice nonpharmaceutical mitigation efforts against SARS-CoV-2.
The ability to be an excellent infection preventionist requires lifelong learning and taking the initiative to grow professionally.
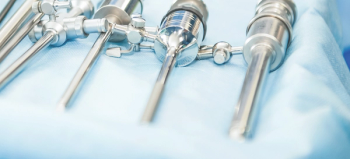
The successful combination of products and the adoption and application of science-based practices will help the sterile processing profession rise above challenges to protecting frontline technicians.

Now is the time for infection preventionists to harness the current attention to biopreparedness and use the momentum to build the foundations for strong local programs that can be sustained through future waves of competing priorities.
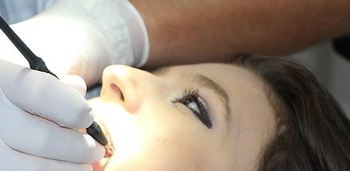
Dental instruments may not seem as invasive as the instruments used in medical surgeries, but they still pose the same dangers.
Having an infection preventionist on site or as a consultant to lead infection prevention and control training makes a difference. The ability to screen, isolate, or group patients can save lives. Ongoing testing of residents and staff is critical.
“[Personal protective equipment] was splashed during most activities and did not prevent skin exposure even when properly donned and doffed,” the study states.

The electrostatic sprayer method kills nearly 100% of pathogens. It also kills the COVID-19 virus. But is that overkill?

As part of infection prevention against COVID-19, schools spent millions of federal dollars trying to upgrade ventilation systems. That money has been ill-spent, warn some experts.

What does the post-COVID-19 future look like for infection preventonists? Great strides in infection prevention have been made because of the COVID-19 response, but look for them to be modified moving forward.
Darrel Hicks: “EVS teams work around professionals who are certified—whether it’s respiratory therapists, physical therapists, the RNs, the doctors—and I think if we ever hoped to elevate their status that we need to certify environmental services workers to a certain level of knowledge before they even start cleaning patient rooms.”
Infection preventionists across health care settings struggle with a myriad of problems during this pandemic. IPs at nursing homes have it particularly hard.
The allocations are scheduled to begin next month, with initial awards totaling $885, of which $500 million will go to what the CDC calls “strike teams” that will focus on nursing homes and other long-term care facilities.

There needs to be a national standard for measuring the effectiveness of personal protective equipment at its most vulnerable spot—the wrist, urge CDC investigators.

Heather Saunders MPH, RN, CIC: “I think [infection preventionists] really need to be aware of what the efforts are at their state health departments and how they can collaborate with those efforts. IPs need to also have their own surveillance systems in place. They need to know what they’re looking for.”

Take 5 minutes to catch up on Infection Control Today®’s highlights for the week ending July 9.