Adenosine Triphosphate (ATP) Bioluminescence Testing and Performance
In 2003, the CDC recommended hospitals to clean and disinfect frequently touched surfaces. Later, they advised hospitals to closely supervise cleaning procedures near patients. This article explains what IPC personnel need to know about ATP testing.
Figure 1 The plate count is the most common method to determine the number of living bacteria on a surface, with results expressed in colony-forming units (CFUs) per surface area tested.
(Adobe Stock photos, unknown)
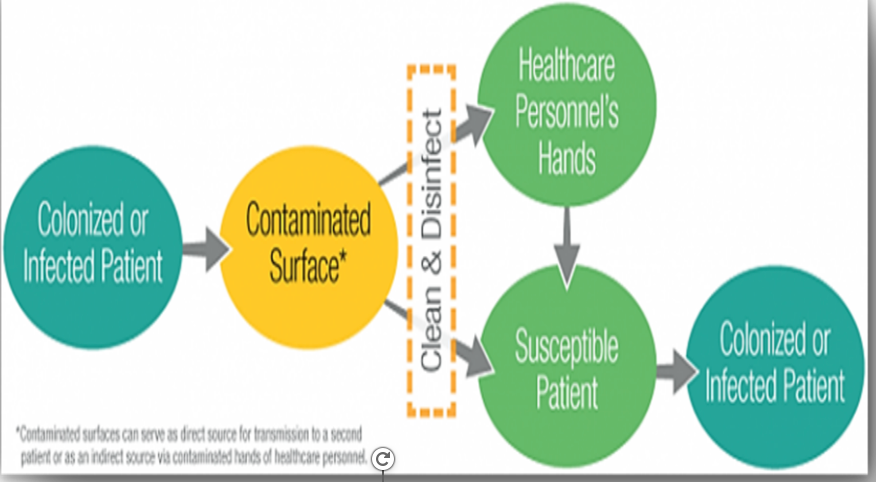
The environment plays a significant role in the transmission of health care-associated pathogens. Environmental cleaning is essential in reducing microbial contamination on hospital surfaces and contributes to the prevention of health care-associated infections (HAIs). Patients infected or colonized with pathogens frequently contaminate their surroundings, which can contribute to the transmission of pathogens by serving as a source that may contaminate the hands or gloves of health care personnel (HCP).1 This can potentially result in cross-contamination of other patients, other patient care items, and other environmental surfaces, as well as self-contamination and possible infection if you touch your face, mouth, or eyes.
Recommendations/Requirements
In 2003, the CDC “Guidelines for Environmental Infection Control in Health care Facilities—Environmental Surfaces” recommended that hospitals clean and disinfect ‘‘high-touch surfaces.’’ A later CDC guideline strongly recommended that hospitals ‘‘monitor (ex supervise and inspect) cleaning performance to ensure consistent cleaning and disinfection of surfaces near the patient and likely to be touched by the patient and health care professionals.’’ As a result of these recommendations, the 2007 revised Center for Medicare and Medicaid Services (CMS) Interpretative Guideline for its infection control standard now requires that hospital infection prevention and control programs ‘‘must include appropriate monitoring of housekeeping activities to ensure that the hospital maintains a sanitary environment.’’2
Reference: diagram-https://www.cdc.gov/hai/prevent/environment/surfaces.html CDC website: Reduce Risk from Surfaces
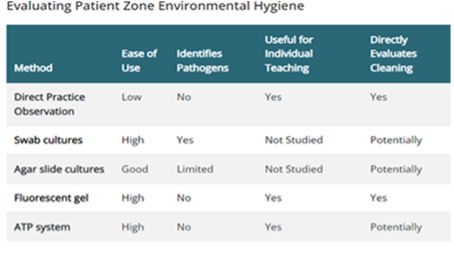
In 2010, the CDC identified 4 science-based methods for evaluating environmental hygiene, including effective surface cleaning. They are direct practice observation, adenosine triphosphate (ATP) bioluminescence, fluorescent markers, swabs, and agar slide cultures.1
ATP Background
More than thirty years ago, the food industry was the first to standardize cleaning methods by choosing ATP testing to assess cleanliness.3 The food industry is highly regulated by the FDA and USDA, as well as other agencies, with specific guidance and established facility cleanliness standards. Besides the food industry, ATP testing has been implemented in other areas, including education, the pharmaceutical industry, electronics manufacturing, and health care.In health care, ATP testing is used to detect residual contamination on hard, non-porous surfaces in the environment, providing an estimate of the presence of organic matter, including microbiological contamination.4 Unfortunately, unlike the food industry, no specific agency or regulatory requirements exist for what constitutes a “clean” surface in health care.
How Does ATP Bioluminescence Technology Work?
ATP is a compound used in the metabolic processes of cells, so it is present in all organic material. ATP bioluminescence technology measures the presence of ATP in organic matter from various sources - living microbes, dead microbes, soil bioburden, and natural materials. The detection of ATP in bioburden is due to an enzymatic reaction resulting in bioluminescence. This reaction utilizes ATP, luciferin, and firefly luciferase, an enzyme that produces light in the firefly's tail.4 During the reaction, luciferin is oxidized and emits light. The light produced during this reaction is related to the amount of ATP in the sample and is reported as “relative light units” (RLU). A device called a luminometer is used to “read” the reaction. However, ATP bioluminescence detection is non-specific. RLU results cannot distinguish between live or dead bacteria, fungi, viruses, or any other intracellular source of ATP, like food or soil residual remaining on a surface.
https://www.cdc.gov/hai/toolkits/appendices-evaluating-environ-cleaning.html
ATP Bioluminescence Pass/Fail Determination
The manufacturers of ATP test systems state that testing does not have one pass number that can apply to every facility and every surface. The amount of ATP on a surface after cleaning and the acceptable RLU limits depends on several factors: 1) the surface material, 2) the surface age, 3) what touched that surface, 4) how often the surface was cleaned, and 5) the cleaning procedure and cleaning elements of including time, temperature, and cleaning chemicals.
To have a robust assessment of the equipment or surfaces being evaluated, rigorous testing with ATP swabs should only be done after a deep cleaning the test surfaces. The testing should be done in several pre-selected testing points as this testing aims to produce data for a defined period (eg 2 to 3 weeks or 10-30 cleaning cycles).5 An analysis of the results should identify a reasonable pass/fail limit for the surface or equipment tested. Each facility should set pass/fail limits based on its own data and risk tolerance for each surface. (Figure 1)
There are no specific standards or regulatory limits on RLU to define what is considered “clean,” a significant limitation of ATP bioluminescence test systems. RLU is not a standardized unit of measurement—different ATP systems have other limits in sensitivities, reagent formulations, swabs, and light detection systems. Every ATP device manufacturer has its own RLU arbitrary scale based on their pre-existing curve of ATP luminescence for that instrument, and all measurements are made relative to that specific system. Because each manufacturer uses a different scale, RLU values should not be used to compare other ATP monitoring systems. This limits comparing data between different ATP units.5
ATP – A Potential Measure of Cleanliness
When deciding on an evaluation method to monitor a cleaning program, it is essential to consider whether the cleaning process or the actual cleanliness of surfaces is the goal. According to the CDC, direct observation and fluorescent gel systems directly evaluate adherence to the cleaning process. In contrast, swab/culture methods and ATP bioluminescence systems have the potential to assess cleanliness by monitoring the effectiveness of cleaning procedures.2 Cleanliness, defined by the Oxford Dictionary, is “the state or quality of being clean or being kept clean.” However, no defined level of microbial contamination correlates with cleanliness in a clinical setting.2 This lack of a standardized threshold or benchmark for determining the level of cleanliness or “Safe” post-cleaning RLU for specific surfaces, equipment, or patient care areas could lead to a feeling of false confidence in the level of contamination on the tested surface.12
ATP bioluminescence detects ATP from bacteria (RLU) in addition to all other biological sources of ATP, so it is not a measure of the number of bacteria on a surface, only a measurement of the material present on the surface. The plate count is the most common method to determine the number of living bacteria on a surface, with results expressed in colony-forming units (CFUs) per surface area tested. CFU and RLU values are detected using different test methods and measure other substances. RLU values do not consistently correlate to CFUs when evaluating an environmental surface. Six studies have shown that various pathogens exhibit different RLU readings.7
ATP - NOT an Assessment of Disinfection
Microbial tests verify the disinfectant is doing its job. Disinfectant efficacy is confirmed by the Environmental Protection Agency (EPA) when a disinfectant meets the criteria to be an EPA-registered disinfectant. ATP bioluminescence testing cannot be trusted to evaluate the disinfection of a health care surface. It should also not be used as a tool to compare the effectiveness of disinfection between different disinfectants. Luminometers have limitations in detecting the actual number of organisms on a surface, which can lead to false confidence in surface disinfection.8 Residual detergents and disinfectants can either increase or decrease RLU readings.2 Additionally, high concentrations of bleach can potentially decrease ATP bioluminescence, reducing RLUs. More research is needed to understand how bleach-based disinfectants impact the use of the ATP test system. Be aware if a bleach-based disinfectant is used, the surface must be dry before testing with ATP. Fortunately, many commercial bioluminescence detection kits contain neutralizers to combat the effect of detergents/sanitizers.9
Contaminants other than bacteria can also interfere with RLU readings. Inconsistent ATP measurements may be caused by microfiber products, manufactured plastics, stainless steel, any organic substrate, or even the material of surfaces being cleaned.7 ATP does not consistently detect bacterial spores. Testing for viral contamination with ATP luminometers is not helpful because viral cells do not contain or produce ATP molecules on their own, raising another concern about the limitations of the ATP bioluminescence technology in health care. Despite these limitations, ATP bioluminescence testing is one of the most common methods used in health care to monitor the effectiveness of environmental cleaning.
Conclusion
When evaluating a method to assess surface cleanliness, understanding the pros and cons of each method should be considered. ATP bioluminescence testing has gained popularity as a method to assess surface cleanliness. However, there are many limitations to this method. ATP performs better than visual inspection but not as well as fluorescent markers.11 There is no correlation between CFU and RLU because ATP does not differentiate between living and dead organisms.6 It only determines the presence of organic material, including dirt and debris. Consider viable plate counts if you want to know the number of viable bacteria on a surface. As mentioned earlier, ATP does not detect viruses or spores consistently.
RLU ranges and sensitivity between commercially available systems vary. ATP testing requires robust testing of surfaces in your facility to generate data so you can set facility-specific pass/fail limits based on facility data and risk tolerance for each surface. However, there are no specific standards or regulatory limitations on RLUs or standards for what is considered “clean.”
Residues of detergents and disinfectants on surfaces, microfiber, and the surface itself can interfere with RLU readings, either increasing or decreasing them.7 ATP bioluminescence testing should not be used to evaluate disinfection of a health care surface. It should not be used as a tool to compare the effectiveness of disinfection between different disinfectants.
ATP bioluminescence testing should be used as a tool to provide education to environmental services and health care personnel on cleaning processes. It can also be used to identify surfaces that might have been missed during a cleaning regimen based on data obtained to determine RLU baselines, but again, there is no standard for what is “clean.” ATP and any other surface cleanliness evaluation method should always be used with good infection control practices, including environmental services education and compliance with cleaning practices and protocols. It is advisable to be aware of the limitations of ATP to use it effectively.
References
- Amodio E, Dino C. Use of ATP bioluminescence for assessing the cleanliness of hospital surfaces: A review of the published literature (1990–2012). DOAJ. March-April 2014. 7(2). 92-98. Doi.org/10.1016/j.jiph.2013.09.005https://www.sciencedirect.com/science/article/pii/S2772431X22000338?via%3Dihub
- Carling P, Bartley J. Evaluating hygienic cleaning in healthcare settings: What you do not know can harm you patients. AJIC. June 2010. 38 (5). 41-50.DOI.org/10.1016/j.ajic.2010.03.004 https://www.ajicjournal.org/article/S0196-6553(10)00406-2/fulltext
- Powitz R. ATP Systems Help Put Clean to the Test. Food Safety Magazine. June 2007. Accessed April 1, 2023. https://www.food-safety.com/articles/4080-atp-systems-help-put-clean-to-the-test
- APIC Practice Guidelines Committee (PGC) Position Statement on Adenosine Triphosphate (ATP) Testing of Reusable Textiles in Healthcare Facilities. August 15, 2012. https://www.apic.org/Resource_/TinyMceFileManager/Position_Statements/APIC_position_ATP_and_reusable_textiles_082012.pdf
- 3M Food Safety News. How clean is it? What ATP can tell you about the safety of your food handling areas. 2023. Accessed April 3, 2023.https://food-safety-news.3m.com/fsn/how-clean-is-it-what-atp-can-tell-you-about-the-safety-of-your-food-handling-areas/
- 3M Clean-Trace Hygiene Management System. ATP, RLUs and CFUs. 3M™ Clean-Trace™ Hygiene Management System website. 2010. Accessed April 2, 2023. https://multimedia.3m.com/mws/media/686753O/clean-trace-atp-rlus-and-cfus.pdf
- Montgomery A, Hammer K, Dubert G, Lo T. Evaluation of adenosine triphosphate bioluminescence-based system in rooms occupied by patients with methicillin-resistant Staphylococcus aureus. IMJ. September 2022.1(3) doi.org/10.1016/j.imj.2022.08.002 https://www.sciencedirect.com/science/article/pii/S2772431X22000338
- Omidbakhsh N, Ahmadpour F, Kenny N. How Reliable Are ATP Bioluminescence Meters in Assessing Decontamination of Environmental Surfaces in Healthcare Settings? IMJ. June 18, 2014. doi.org/10.1371/journal.pone.009995. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0099951#s4
- Tetro J, Sattar S. The Application of ATP Bioluminescence for Rapid Monitoring of Microbiological Contamination on Environmental Surfaces: A Critical Review By Jason "The Germ Guy". Infection Control.tips. November 10, 2021. Accessed April 3, 2023. https://infectioncontrol.tips/2021/11/10/atp-critical-review
- Options for Evaluating Environmental Cleaning. Appendix B. CDC. Updated May 16, 2019. Accessed April 4, 2023. https://www.cdc.gov/hai/toolkits/appendices-evaluating-environ-cleaning.html
- Rutala W, Kanamori H, Gergen M, Sickbert-Bennett E, Huslage K, Weber D. Comparative Analysis of Four Major Hospital Cleaning Validation Methods. AJIC. 2017. 4(87)doi.org/10.1016/j.ajic.2017.04.087https://www.ajicjournal.org/article/S0196-6553(17)30405-4/fulltext
- Best Practices for Environmental Cleaning in Healthcare Facilities: in Resource-Limited Settings. CDC. 2019. Version 2. Accessed April 10, 2023. https://www.cdc.gov/hai/pdfs/resource-limited/environmental-cleaning-RLS-H.pdf
Newsletter
Stay prepared and protected with Infection Control Today's newsletter, delivering essential updates, best practices, and expert insights for infection preventionists.
Reducing Hidden Risks: Why Sharps Injuries Still Go Unreported
July 18th 2025Despite being a well-known occupational hazard, sharps injuries continue to occur in health care facilities and are often underreported, underestimated, and inadequately addressed. A recent interview with sharps safety advocate Amanda Heitman, BSN, RN, CNOR, a perioperative educational consultant, reveals why change is overdue and what new tools and guidance can help.
New Study Explores Oral Vancomycin to Prevent C difficile Recurrence, But Questions Remain
July 17th 2025A new clinical trial explores the use of low-dose oral vancomycin to prevent Clostridioides difficile recurrence in high-risk patients taking antibiotics. While the data suggest a possible benefit, the findings stop short of statistical significance and raise red flags about vancomycin-resistant Enterococcus (VRE), underscoring the delicate balance between prevention and antimicrobial stewardship.
What Lies Beneath: Why Borescopes Are Essential for Verifying Surgical Instrument Cleanliness
July 16th 2025Despite their smooth, polished exteriors, surgical instruments often harbor dangerous contaminants deep inside their lumens. At the HSPA25 and APIC25 conferences, Cori L. Ofstead, MSPH, and her colleagues revealed why borescopes are an indispensable tool for sterile processing teams, offering the only reliable way to verify internal cleanliness and improve sterile processing effectiveness to prevent patient harm.
The Next Frontier in Infection Control: AI-Driven Operating Rooms
Published: July 15th 2025 | Updated: July 15th 2025Discover how AI-powered sensors, smart surveillance, and advanced analytics are revolutionizing infection prevention in the OR. Herman DeBoard, PhD, discusses how these technologies safeguard sterile fields, reduce SSIs, and help hospitals balance operational efficiency with patient safety.
Targeting Uncertainty: Why Pregnancy May Be the Best Time to Build Vaccine Confidence
July 15th 2025New national survey data reveal high uncertainty among pregnant individuals—especially first-time parents—about vaccinating their future children, underscoring the value of proactive engagement to strengthen infection prevention.