Parker’s High-Level Disinfectant Foam Receives FDA de novo Clearance
Neal Buchalter, MBA, president of Parker Laboratories, spoke with Infection Control Today® in an exclusive interview about the product, the companies involved, and how it came to the US market.
Neal Buchalter, MBA, president of Parker Laboratories
(Photo courtesy of Parker Laboratories)

Chlorine dioxide is a safe and effective disinfectant, widely used in disinfecting drinking water, as noted in a 2022 study published in the Journal of Medicine and Life. In June 2023, at the Association for Professionals in Infection Control and Epidemiology (APIC) Annual Conference and Exposition in Orlando, Florida, Parker Laboratories and Tristel plc presented their new foam with chlorine dioxide.
Parker Laboratories had recently received clearance from the FDA for Tristel ULT, a disinfecting foam containing chlorine dioxide that can be used as a high-level disinfectant for ultrasound probes utilized in body cavities and skin-surface transducers. This unique foam is exclusively manufactured and distributed by Parker Laboratories for US markets in partnership with UK-based Tristel plc. Tristel ULT is the only high-level disinfectant foam safe for ultrasound probes and has been proven effective in killing pathogens such as SARS-CoV-2 and Mycobacterium tuberculosis.
During a recent exclusive interview with Infection Control Today® (ICT®), Neal Buchalter, MBA, president of Parker Laboratories, provided insight into the company’s product and its introduction to the US market. Additionally, he explained the significance of obtaining a de novo clearance.
ICT: Please describe your product, what this disinfecting foam is, why it’s unique, and what it does.
Neal Buchalter, MBA: Tristel ULT is a product that, in one form or another, has been available in Europe, Australia, and a few other markets for many years and is now being introduced into the United States through Parker Laboratories. It is a chlorine dioxide foam, cleared for use as a high-level disinfectant.
What is unique about that is it’s point-of-care high-level disinfection. It doesn’t require equipment; it doesn’t require ventilation, plumbing, or maintenance contracts. It’s strictly supply-based. And outside of old glutaraldehyde soaks, which, hopefully, nobody uses, there’s nothing like that on the market today.
ICT: When you say point of care, you don’t mean at the bedside; you mean in the sterile processing department, correct?
NB: No, we mean in the ultrasound exam room. I don’t know if I’d have it next to a patient, but it can be utilized in an exam room.
The product is distributed in a dual-chambered pump bottle that contains 2 different chemicals, a base, and an activator. When the user depresses the pump, the 2 chemicals combine to generate a consistent dose of chlorine dioxide foam. The foam is very safe to handle. Users should wear typical PPE [personal protective equipment], but the foam is quite safe and is considered to be, at most, a mild skin irritant and a mild eye irritant.
Tristel Ult dosing picture.
(Photo courtesy of Parker Laboratories)
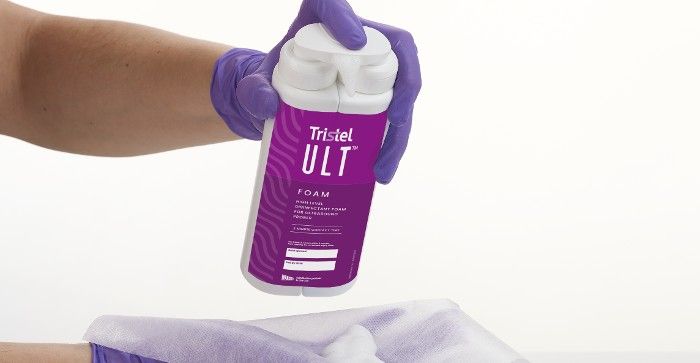
Tristel ULT comes with a specific nonabrasive, lint-free, nonabsorbent, dry wipe validated for use with the foam. The user dispenses 4 doses of the foam onto a wipe, waits 10 seconds for the chemicals to activate, wipes the transducer 4 times, and then lets the transducer sit for 2 minutes. At the end of 2 minutes, the user takes another wipe to remove any remaining solution from the transducer. That’s the end of the process, and you have high-level disinfection of ultrasound devices.
ICT: There’s no water, so there can’t be any water contamination at all.
NB: There’s no water. It’s high-level disinfection [HLD].
You do need a cleaning step before disinfection. There are lots of products to use for that. One of them is our Protex low-level disinfection wipes. You can also use soap and water if you choose. That’s the type of cleaning step that is done with all other HLD equipment as well.
ICT: That makes sense. Usually, that thing would have to go down to the sterile processing unit or department, and then you have that turnaround time.
NB: Correct. You take the transducer out of circulation. And these are busy practices, so nobody wants to do that.
ICT: And I would assume, by the way you’re describing this, it wouldn’t have to be someone certified in sterile processing to do the cleaning.
NB: No, I would hesitate because that sounds like hospital protocol. Perhaps, but it’s certainly not necessary. A technician can do it.
ICT: What is an FDA de novo classification? What specifically did you have to do to get that classification?
NB: I can talk about this in generalities. We are a partner of the UK company Tristel plc, and we are licensed to manufacture and distribute Tristel ULT in North America. We didn’t do the regulatory for the product; Tristel did. They originally pursued a premarket notification (510(k)), but there is no predicate device, so they needed to follow the FDA’s de novo process, which is much more extensive and requires a much higher level of testing and validation.
Tristel performed efficacy studies and clinical studies with clinical equipment in use. They also performed usability studies, where they had to validate the product’s instructions for use (IFUs). They had untrained users read the IFUs and go through the steps to ensure that users could perform them accurately. So the requirements are much more stringent than a 510(k).
At the end of the de novo process, FDA creates a new Class II category for the product that passed through the process. For Tristel ULT, the new category that the agency created has the generic name “foam or gel chemical sterilant/high-level disinfectant.” And FDA describes products in the new category as “a foam or gel chemical sterilant/high-level disinfectant is a germicide in the form of a foam or gel intended for use as the terminal step in high-level disinfection of medical devices prior to patient use.” It took a long time, but they’ve done an excellent job, and they just got clearance at the beginning of June.
ICT: How long has it been used overseas?
NB: At least 10 years, in various forms. Not exactly the packaging we’re using today but in different forms. Talking to Tristel reps at APIC, they said the product had been applied more than a hundred million times in other markets. Because the product is somewhat mature in other markets, it is already listed as approved for use on nearly a thousand different ultrasound transducers and probes. So we don’t have to go through the process of building that dataset. A lot of that already exists.
FDA granted its clearance in the first couple of days of June. We plan to make the product available on October 1, so it’s not on the US market yet. We’re very excited about it. The markets we feel it will be most widely used in are OB/GYN and urology, as well as the growing vascular access market.
There’s been an ongoing debate about what level of disinfection is necessary for percutaneous procedures. We’re supportive of the Intersocietal Position Statement developed under the leadership of the American Institute for Ultrasound in Medicine (AIUM), which effectively says that intermediate-level disinfection capable of killing tuberculosis is the minimum requirement.
But we know that many hospitals also want the option of a fast and convenient high-level disinfectant. These are fast-moving departments. Many of these procedures are done in the emergency department. They need a point-of-care solution, which is an excellent solution for them.
The existing products on the market don’t fit the needs of some of these clinicians. We feel this product is superior to those already on the market for the reasons I’ve already cited: that it’s point-of-care, that the kill time is just 2 minutes, and that it’s easy for clinicians to use and get back to patient care.
ICT: When I speak to people about their products, my first question is, how easy is it to use? Because for many reasons, we already don’t have the personnel we need, so we don’t want to pull people away. We need to have products that are easy to use, can be done, and then get out. We need to have products that are easy to use.
NB: One other point that’s very important to hospitals is the ability to track and trace the performance of high-level disinfection. When you have a manual process such as this, you potentially lose the ability to track and trace. We provide logbooks where users can record their cleaning and disinfection operations, but we recognize that these can be cumbersome.
Tristel has developed a very elegant option in the form of an app-based track and trace system to be used in conjunction with the product. The app will be available as a free download for smartphones and tablets, so hospitals can choose whatever equipment platform works best for their environments.
In practice, the user scans the transducer or probe, scans the patient’s wristband or barcode, scans their clinician ID, and then the app will walk them through each process step.
So it tells you to spray the 4 doses. You spray the 4 doses. You wave your hand over the device, and it goes to the next step to wait 10 seconds for the chemical to activate. Again, you wave your hand over the device after it counts down, and then it prompts you to wipe the transducer and wait 2 minutes, and then you can conclude the steps within the device. The app will log and retain all of that activity for hospital administrators. We demonstrated the app at APIC (conference], and the clinicians were pleased to see that functionality.
Additionally, there’s no capital equipment purchase, no maintenance contract, and no necessary test strips—all of which can be quite expensive.
Because the active chemicals of Tristel ULT are blended at the time of use, FDA’s de novo clearance does not require clinicians to perform any separate testing using biological indicators or other test strips. The agency did require that a testing method be made available, so a test strip and device can be used to validate the product's efficacy. But you can’t test every dose because you will use those doses. If a hospital wants to validate the efficacy of the Tristel ULT foam, we recommend that staff test each bottle on the first dose or one dose at the beginning of every day or work shift. But the decision to do so is entirely up to the hospital; there’s no requirement for such testing.
We estimate the cost of using competitive products at roughly $7 per application. By contrast, we expect the cost of using Tristel ULT will be between $2.50 and $3 per application. On top of that, using Tristel ULT will eliminate the disruptive effects of dealing with capital equipment used for high-level disinfection—purchase costs, calibration, maintenance, and the time required to transport and retrieve transducers between uses. So we’re excited about the product positioning for Tristel ULT.
ICT: How did the 2 companies come together to bring it to the United States?
NB: I’ve known this company and the CEO for a long time, and they were never interested in tackling the US market because of the regulatory effort required. But he reached out to me when they decided to invest in it.
In the United States, chlorine dioxide is primarily used in water systems. It’s used in some fumigation processes and for some types of industrial disinfection. I know it’s used in the paper industry, but it’s never been in the medical field.
In some of these other countries, chlorine dioxide is used in other areas of disinfection, so the regulatory concern or regulatory effort needed is vastly different. That’s why they took so long to decide to come here.
ICT: Where did they get the idea?
NB: There’s a long history of development for Tristel ULT as well. Early on, the company did a little work directed specifically toward ultrasound devices, but then they went in other directions before coming back to ultrasound.
Broadening the Path: Diverse Educational Routes Into Infection Prevention Careers
July 4th 2025Once dominated by nurses, infection prevention now welcomes professionals from public health, lab science, and respiratory therapy—each bringing unique expertise that strengthens patient safety and IPC programs.
How Contaminated Is Your Stretcher? The Hidden Risks on Hospital Wheels
July 3rd 2025Despite routine disinfection, hospital surfaces, such as stretchers, remain reservoirs for harmful microbes, according to several recent studies. From high-touch areas to damaged mattresses and the effectiveness of antimicrobial coatings, researchers continue to uncover persistent risks in environmental hygiene, highlighting the critical need for innovative, continuous disinfection strategies in health care settings.