
News





New research at Harvard explains how bacterial biofilms expand to form slimy mats on teeth, pipes, surgical instruments and crops. Through experiment and mathematical analysis, researchers have shown that the extracellular matrix (ECM), a mesh of proteins and sugars that can form outside bacterial cells, creates osmotic pressure that forces biofilms to swell and spread. The ECM mechanism is so powerful that it can increase the radius of some biofilms five-fold within 24 hours. The results have been published in the Proceedings of the National Academy of Science.





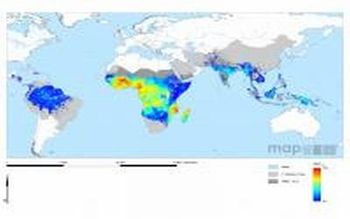
A new suite of malaria maps has revealed in unprecedented detail the current global pattern of the disease, allowing researchers to see how malaria has changed over a number of years.





A recently published study investigating hepatitis B vaccination rates in the United States found that more than half of adults at risk for hepatitis B virus remain unvaccinated. With many of these individuals making contact with the healthcare system, including HIV testing, this statistic reflects many missed opportunities to vaccinate this population.







Researchers at the University of Maryland's A. James Clark School of Engineering, backed by funding from the Robert W. Deutsch Foundation, are developing tools that promise powerful new ways to combat catheter-based and other infections without provoking bacterial resistance to antibiotics.




